Bexion Pharmaceuticals, Inc. to Present at the 2021 Discovery on Target Conference
FOR IMMEDIATE RELEASE [Covington, KY—September 17, 2021]
Bexion Pharmaceuticals, Inc., a clinical-stage biopharmaceutical company, announced today that Gilles Tapolsky PhD, MBA, Bexion’s Vice President, Pharmacology will be virtually presenting preclinical and clinical data of the Company’s lead compound, BXQ-350. The presentation will be made at the Discovery on Target (DOT) Conference in the Immunology and Oncology- Emerging Drug Targets Segment:
BXQ-350: Stimulating the Immune System by Modulating Phosphatidylserine Expression and Sphingolipid Signaling
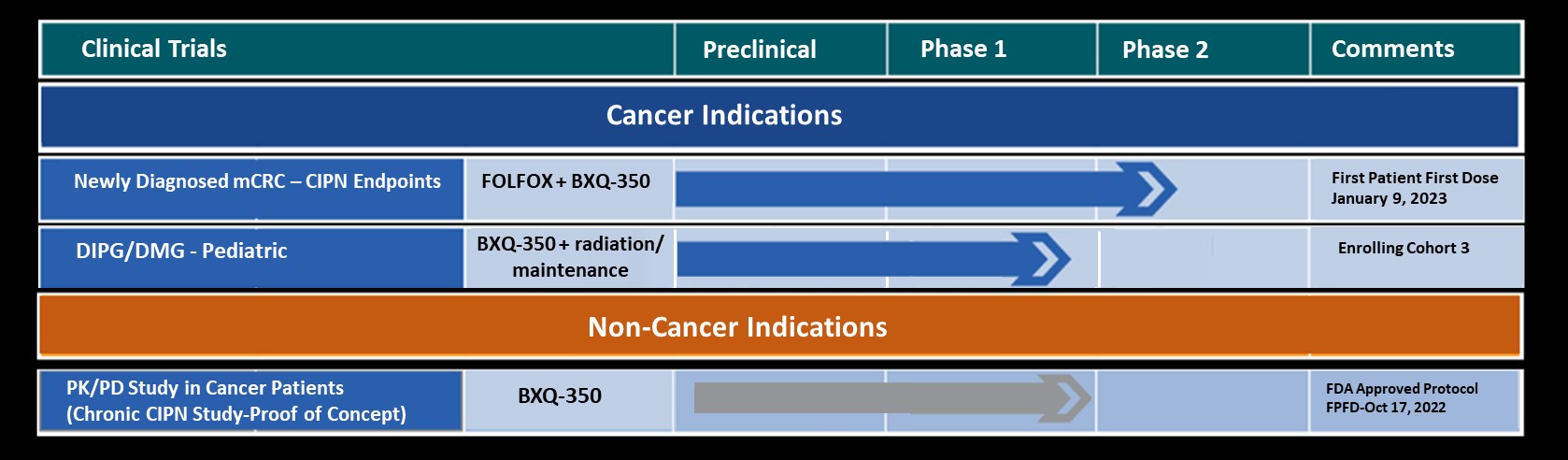
BXQ-350, a nanovesicle formulation of Saposin C, targets phosphatidylserine and modulates sphingolipid metabolism and de novo biosynthesis of ceramides. Expression of phosphatidylserine has been recognized as a global immunosuppressive signal and ceramides are important cell signaling and immune-stimulating molecules. Results of Phase 1 studies in cancer patients demonstrated that BXQ-350 was safe and well-tolerated and had preliminary evidence of single agent activity in some patients.
DOT is the industry’s preeminent event on novel drug targets and technologies for drug discovery professionals. DOT highlights advances in current and emerging “hot” targets and technologies, as well as target validation strategies for the discovery and development of novel therapeutic agents, ranging from biologics to small molecules. The meeting will be held September 27-30, 2021.
About Bexion Pharmaceuticals
Bexion Pharmaceuticals, a clinical-stage biopharmaceutical company, is pioneering the development of life-changing treatments by leveraging the untapped mechanisms of the lysosome. Bexion believes the lysosome is an underexploited cellular orchestrator involved in multiple diseases. Bexion’s lead drug candidate is BXQ-350, a first-in-class biologic containing the multifunctional, lysosomal activator protein, Saposin C and a phosphatidylserine.
BXQ-350 has demonstrated pre-clinical antitumor effects in vitro and in vivo, particularly in brain and other solid tumors, including those that may lead to brain metastases. Bexion has completed a multi-site first-in-human Phase 1 clinical trial of BXQ-350 for solid tumors and gliomas. Bexion is in Phase 1/2 for a rare pediatric brain tumor and plans to initiate two adult Phase 2 programs in 2022.
Media Contact: Margaret van Gilse ●859.757.1652 ● [email protected].
Forward-Looking Statements
This press release contains forward-looking statements made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995 that involve risks, uncertainties and assumptions that could cause Bexion’s actual results and experience to differ materially from anticipated results and expectations expressed in these forward-looking statements. Bexion has in some cases identified forward-looking statements by using words such as “anticipates,” “believes,” “hopes,” “estimates,” “looks,” “expects,” “plans,” “intends,” “goal,” “potential,” “may,” “suggest,” and similar expressions. Among other factors that could cause actual results to differ materially from those expressed in forward-looking statements are Bexion’s need for, and the availability of, substantial capital in the future to fund its operations and research and development; the fact that Bexion’s compounds may not successfully complete pre-clinical or clinical testing, or be granted regulatory approval to be sold and marketed in the United States or elsewhere. You should not place undue reliance on any forward-looking statements. Bexion undertakes no obligation to release publicly the results of any revisions to any such forward-looking statements that may be made to reflect events or circumstances after the date of this press release or to reflect the occurrence of unanticipated events, except as required by applicable law or regulation.