Bexion Pharmaceuticals, Inc. Announces Poster Presentation at ASCO GI 2024
COVINGTON, Ky., Jan. 16, 2024 /PRNewswire/ — Bexion Pharmaceuticals, Inc., a clinical-stage biopharmaceutical company developing a new generation of biologic therapy to treat solid tumor cancers and chemotherapy-induced peripheral neuropathy (CIPN), today announced a Trials in Progress poster presentation at the American Society of Clinical Oncology Gastrointestinal Cancers Symposium (ASCO GI), being held January 18-20, 2024, in San Francisco, CA. Poster details are included below.
Poster Details:
Session Title: Trials in Progress Poster Session C: Cancers of the Colon, Rectum, and Anus
Session Date: January 20th, 2024
Abstract Number: TPS224
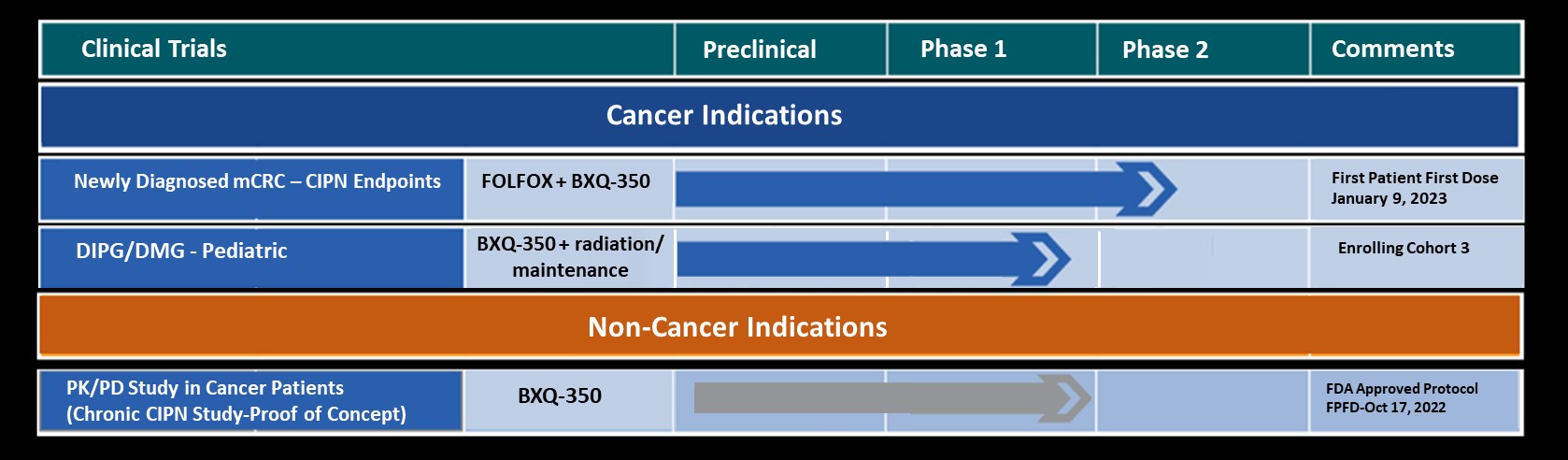
Abstract Title: “BXQ-350: A phase 1b/2 placebo controlled, double blinded study on the efficacy and safety of BXQ-350 in combination with mFOLFOX7 and bevacizumab in newly diagnosed metastatic colorectal carcinoma (mCRC).”
“We are excited to present our ongoing trial of BXQ-350 in mCRC during the 2024 ASCO GI Symposium” said Scott Shively, CEO and President of Bexion Pharmaceuticals. “BXQ-350 in combination with FOLFOX and bevacizumab offers a unique opportunity to improve outcomes for 1st line patients with mCRC. We look forward to sharing progress with investigators and leaders at the conference.”
About BXQ-350
Bexion’s lead drug candidate is BXQ-350, a first-in-class biologic containing the multifunctional, sphingolipid activator protein, Saposin C, and a phospholipid. BXQ-350 has pre-clinical antitumor effects in vitro and in vivo, particularly in colorectal, brain and other solid tumors. Two Phase 1 clinical trials, one in adults and one in pediatric DIPG patients, demonstrated a strong safety profile for BXQ-350 with evidence of single agent activity across multiple solid tumors. Additionally, other clinical and non-clinical data suggest BXQ-350 has activity in chemotherapy-induced peripheral neuropathy.
About Bexion Pharmaceuticals
Bexion Pharmaceuticals, a clinical-stage biopharmaceutical company, is developing a new generation of biologic therapy to treat solid tumor cancers and chemotherapy-induced peripheral neuropathy (CIPN) with potential portfolio expansion opportunities in other cancers and broader neuropathic pain indications. Bexion currently has three clinical trials open for enrollment.
Investor Contact:
William Windham
Solebury Strategic Communications
646-378-2946
[email protected]
Media Contact:
Joyce LaViscount
Bexion Pharmaceuticals
859-446-7386
[email protected]