Bexion Pharmaceuticals, Inc. Presents Clinical Data at 2018 SNO Annual Meeting
FOR IMMEDIATE RELEASE Covington, KY ~ 19 November 2018
Bexion Pharmaceuticals, Inc., a clinical-stage biopharmaceutical company focused on rare brain and solid tumors, announced today that a clinical oral report followed by poster review and a second case study poster were presented at the Society of Neuro-Oncology (SNO) Annual Meeting held November 16-18 in New Orleans, LA. The SNO Annual Meeting brings together more than 2600 researchers and clinicians in the field of neuro-oncology.
Bexion’s representation:
High-Grade Glioma Outcomes in the Phase 1 BXQ-350 Trial of Cancer-Selective SapC-DOPS Nanovesicles
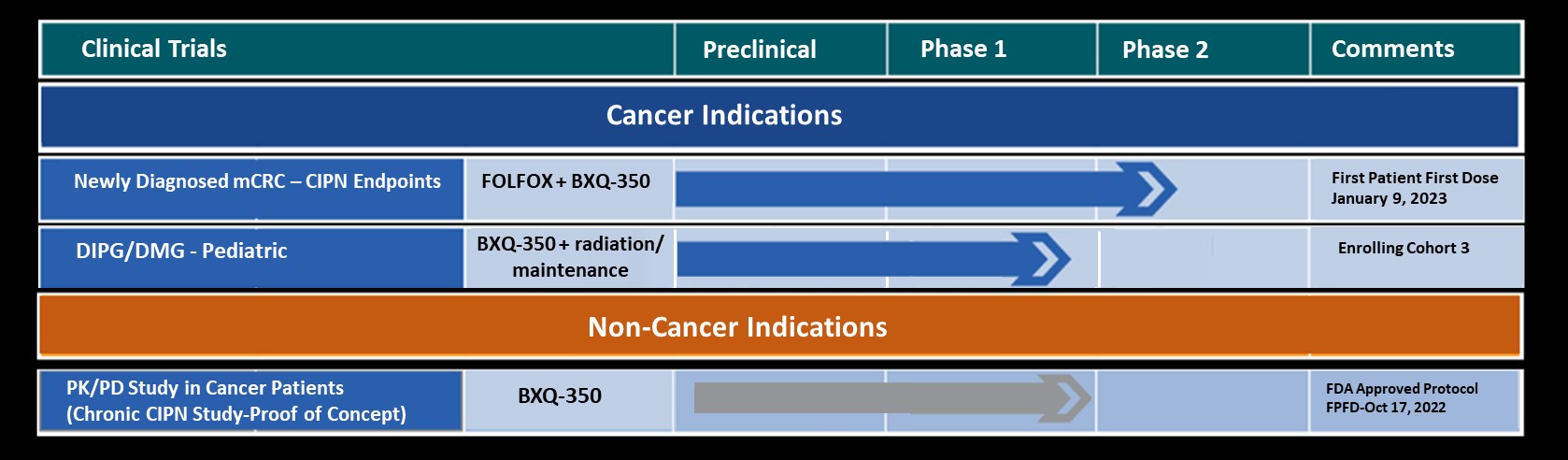
Vinay Puduvalli, MD, Professor and Director, Division of Neuro-Oncology, The Ohio State University Comprehensive Cancer Center gave an oral presentation on November 16th discussing the safety outcomes of High Grade Glioma (HHG) patients from a Phase 1a BXQ-350 trial (NCT02859857). This was followed by a traditional poster viewing.
Initial Experience in Ependymoma with Investigational Cancer-Targeting BXQ-350 SapC-DOPS Nanovesicles: A Rare Tumor Case Study
During a poster presentation on Saturday November 17th, John Villano MD, Director, Clinical Neuro-Oncology Research Program, Markey Cancer Center and Professor of Medicine, University of Kentucky HealthCare discussed the safety profile in a rare tumor type resulting from a Phase 1a BXQ-350 trial (NCT02859857).
“Bexion was honored to participate in the SNO Annual meeting. We are excited to have presented new BXQ-350 data from our Phase 1 program by our Principal Investigators,” stated Dr. Ray Takigiku, Founder and CEO of Bexion.
About BXQ-350
BXQ-350 is a unique formulation of a synthetically produced, human lysosomal protein, Saposin C (sphingolipid activator protein, or SapC), and the phospholipid dioleoylphosphatidylserine (DOPS).
About Bexion Pharmaceuticals
Bexion Pharmaceuticals, a clinical-stage biopharmaceutical company is developing BXQ-350, a first-in-class agent composed of the multifunctional, lysosomal activator protein Saposin C and phosphatidylserine, that has demonstrated antitumor effects in vitro and in vivo, particularly in brain and other solid tumors, including those that may lead to brain metastases. Bexion has completed a multi-site first-in-human, Phase 1a study of BXQ-350 for solid tumors and gliomas and is currently enrolling patients in an expansion phase.
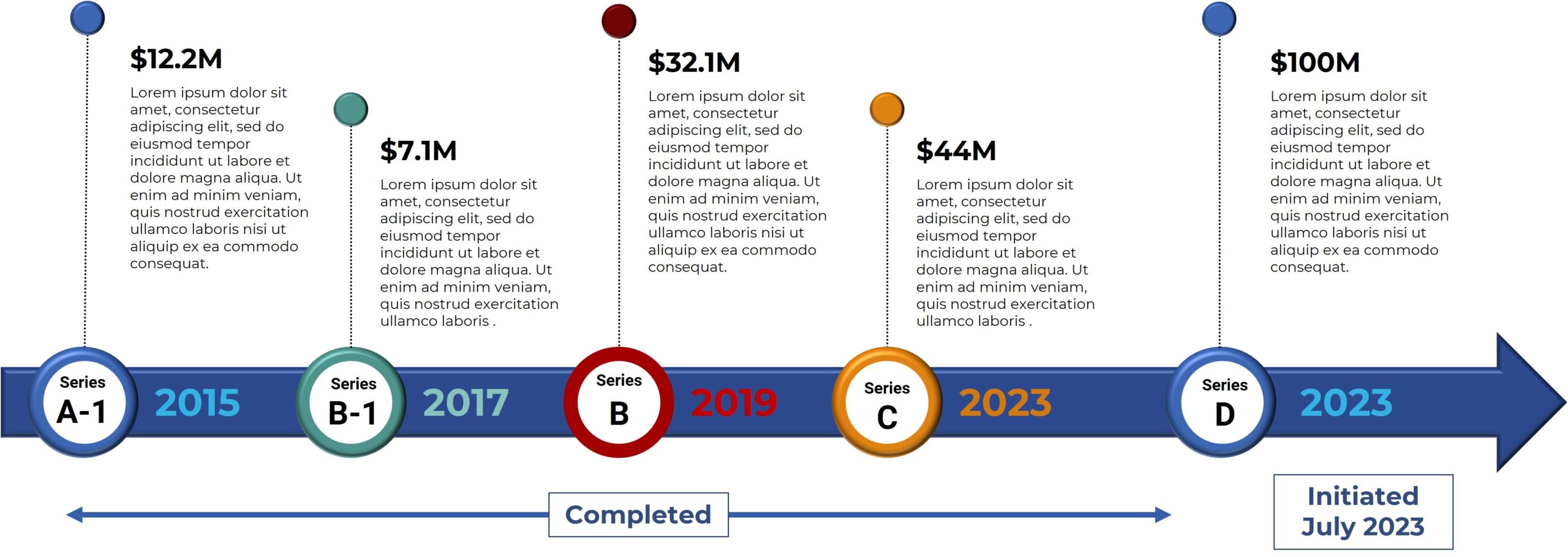
Bexion has research programs in other pathway-related therapeutic areas, including CNS diseases. Currently Bexion is in a Series B Fund Raise. For more information, visit bexionpharma.dev.neptuneweb.com
About SNO
The Society for Neuro-Oncology is a multidisciplinary organization dedicated to promoting advances in neuro-oncology through research and education. Now in its twenty fourth year, the Society continues to grow and mature as the premier North American organization for clinicians, basic scientists, nurses and other health care professionals whose focus is central nervous system tumors in children and adults.
Media Contact: Margaret van Gilse ●859.757.1652 ● [email protected].