Dr. Ray Takigiku Honored as Finalist at the 2019 Healthcare Hero Awards
FOR IMMEDIATE RELEASE Covington, KY – 22 February 2019
Dr. Ray Takigiku, Founder and CEO of Bexion Pharmaceuticals, Inc. was honored as a finalist in the category of “Entrepreneur” of the Year by the Cincinnati Business Courier at the 2019 Healthcare Hero Awards Celebration. These annual awards honor individuals and organizations that have improved the quality of health care in Greater Cincinnati through medical expertise, innovative approaches, management skills, entrepreneurial initiatives, community outreach and patient care.
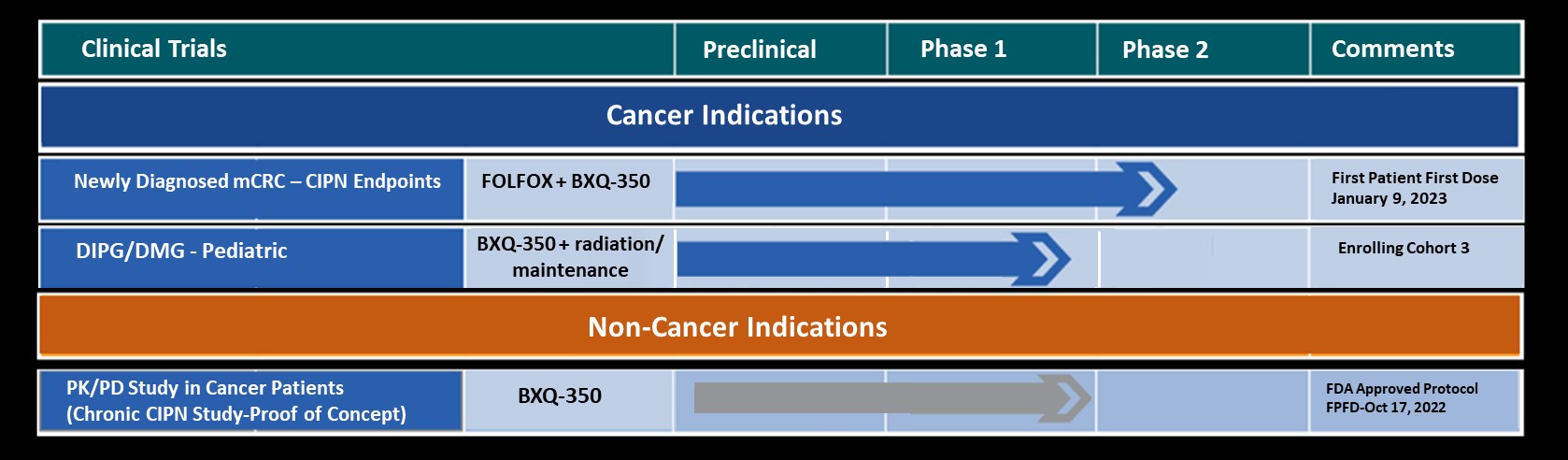
In 2006 Dr. Takigiku founded Bexion Pharmaceuticals, based on a technology platform licensed from Cincinnati Children’s Hospital Medical Center (CCHMC). Under Dr. Takigiku’s leadership, Bexion has progressed their lead compound, BXQ-350, from early pre-clinical animal studies to dosing their first patients in a Phase I Safety Trial for the treatment of cancer. Preliminary data from the Phase I study support a very safe and tolerable drug profile.
“It is an honor to be recognized by the Cincinnati Business Courier and the local community for this award,” stated Dr. Takigiku. “After many years of perseverance, focused commitment and hard work, we have taken this accidental discovery at CCHMC and developed it into a cancer drug that is now in the clinic.”
Dr. Takigiku and his team are aggressively expanding their clinical program with the addition of a Pediatric Phase I Safety study. The first patient is planned to be dosed at CCHMC in late March.
Prior to founding Bexion, Dr. Takigiku was Director of Discovery Core Technologies, Analytical and Bioanalytical for Procter & Gamble Pharmaceuticals, and Interim Co-Director of the Genome Research Institute (now Metabolic Diseases Institute) of the College of Medicine, University of Cincinnati.
About Bexion Pharmaceuticals
Bexion Pharmaceuticals is a clinical-stage biopharmaceutical company developing BXQ-350, a first-in-class agent composed of the multifunctional, lysosomal activator protein Saposin C and phosphatidylserine. BXQ-350 has demonstrated pre-clinical antitumor effects in vitro and in vivo, particularly in brain and other solid tumors, including those that may lead to brain metastases. Bexion has completed multi-site first-in-human Phase 1 Parts 1 and 2 clinical studies of BXQ-350 for solid tumors and gliomas. A Phase 1 Part 3 expansion phase study is currently enrolling patients.
For more information, visit bexionpharma.dev.neptuneweb.com.
Media Contact: Margaret van Gilse ●859.757.1652 ● [email protected].
Forward-Looking Statements
This press release contains forward-looking statements made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995 that involve risks, uncertainties and assumptions that could cause Bexion’s actual results and experience to differ materially from anticipated results and expectations expressed in these forward-looking statements. Bexion has in some cases identified forward-looking statements by using words such as “anticipates,” “believes,” “hopes,” “estimates,” “looks,” “expects,” “plans,” “intends,” “goal,” “potential,” “may,” “suggest,” and similar expressions. Among other factors that could cause actual results to differ materially from those expressed in forward-looking statements are Bexion’s need for, and the availability of, substantial capital in the future to fund its operations and research and development; the fact that Bexion’s compounds may not successfully complete pre-clinical or clinical testing or be granted regulatory approval to be sold and marketed in the United States or elsewhere. You should not place undue reliance on any forward-looking statements. Bexion undertakes no obligation to release publicly the results of any revisions to any such forward-looking statements that may be made to reflect events or circumstances after the date of this press release or to reflect the occurrence of unanticipated events, except as required by applicable law or regulation.