Bexion Pharmaceuticals, Inc. Receives Rare Pediatric Disease Designation
FOR IMMEDIATE RELEASE [Covington, KY] September 30, 2020
Bexion Pharmaceuticals, Inc., a clinical stage biotechnology company focused on developing innovative cures for cancer, announced that the Federal Drug Administration (FDA) has granted the company’s request for rare pediatric disease designation for BXQ-350, Bexion’s lead therapeutic, for the treatment of diffuse intrinsic pontine glioma (DIPG) and diffuse midline glioma (DMG).
Rare Pediatric Disease designation is granted for diseases that primarily affect children ages 18 years or younger and fewer than 200,000 people in the U.S. Upon approval of BXQ-350 for a rare pediatric disease in the U.S., Bexion is eligible for a Rare Pediatric Disease Priority Review Voucher, which may be redeemed for priority review of a subsequent product, transferred, or sold. The program is intended to encourage development of new treatments for rare pediatric diseases.
“Achieving this designation for BXQ-350 in treating DIPG/DMG is pivotal to our clinical strategy,” stated Dr. Ray Takigiku, CEO of Bexion. “This first step could lead to a rare pediatric disease priority review voucher upon marketing approval of BXQ-350 for DIPG/DMG.”
About Bexion Pharmaceuticals
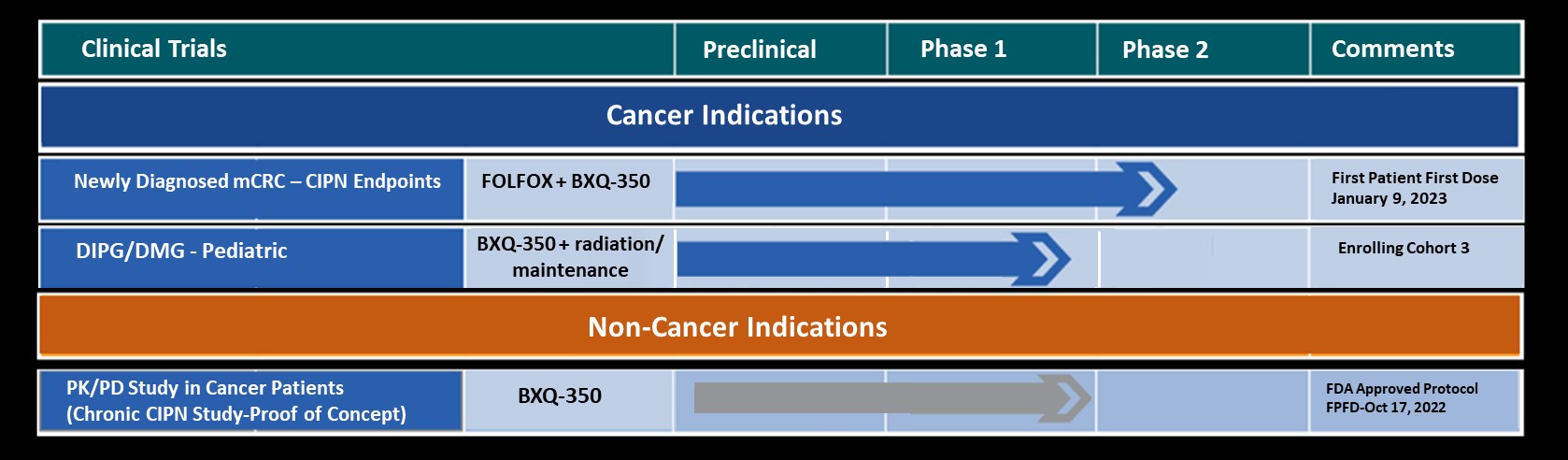
Bexion Pharmaceuticals is a clinical-stage biopharmaceutical company developing BXQ-350, a first-in-class agent composed of the multifunctional, lysosomal activator protein Saposin C and phosphatidylserine. BXQ-350 has demonstrated pre-clinical antitumor effects in vitro and in vivo, particularly in brain and other solid tumors, including those that may lead to brain metastases. Bexion has completed a multi-site first-in-human Phase 1 clinical trial of BXQ-350 for solid tumors and gliomas. A Phase 1 Pediatric Trial enrollment was completed in October, 2019.
Media Contact: Margaret van Gilse ●859.757.1652 ● [email protected].
Forward-Looking Statements
This press release contains forward-looking statements made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995 that involve risks, uncertainties and assumptions that could cause Bexion’s actual results and experience to differ materially from anticipated results and expectations expressed in these forward looking statements. Bexion has in some cases identified forward-looking statements by using words such as “anticipates,” “believes,” “hopes,” “estimates,” “looks,” “expects,” “plans,” “intends,” “goal,” “potential,” “may,” “suggest,” and similar expressions. Among other factors that could cause actual results to differ materially from those expressed in forward-looking statements are Bexion’s need for, and the availability of, substantial capital in the future to fund its operations and research and development; the fact that Bexion’s compounds may not successfully complete pre-clinical or clinical testing, or be granted regulatory approval to be sold and marketed in the United States or elsewhere. You should not place undue reliance on any forward-looking statements. Bexion undertakes no obligation to release publicly the results of any revisions to any such forward-looking statements that may be made to reflect events or circumstances after the date of this press release or to reflect the occurrence of unanticipated events, except as required by applicable law or regulation.